The BPC-157 research benefits for tendon repair are a major focus of regenerative medicine in 2026. This stable gastric pentadecapeptide is being studied for its ability to accelerate healing in musculoskeletal tissues. If you are looking at peptide stacks for skin repair, BPC-157 is often the primary compound of interest.
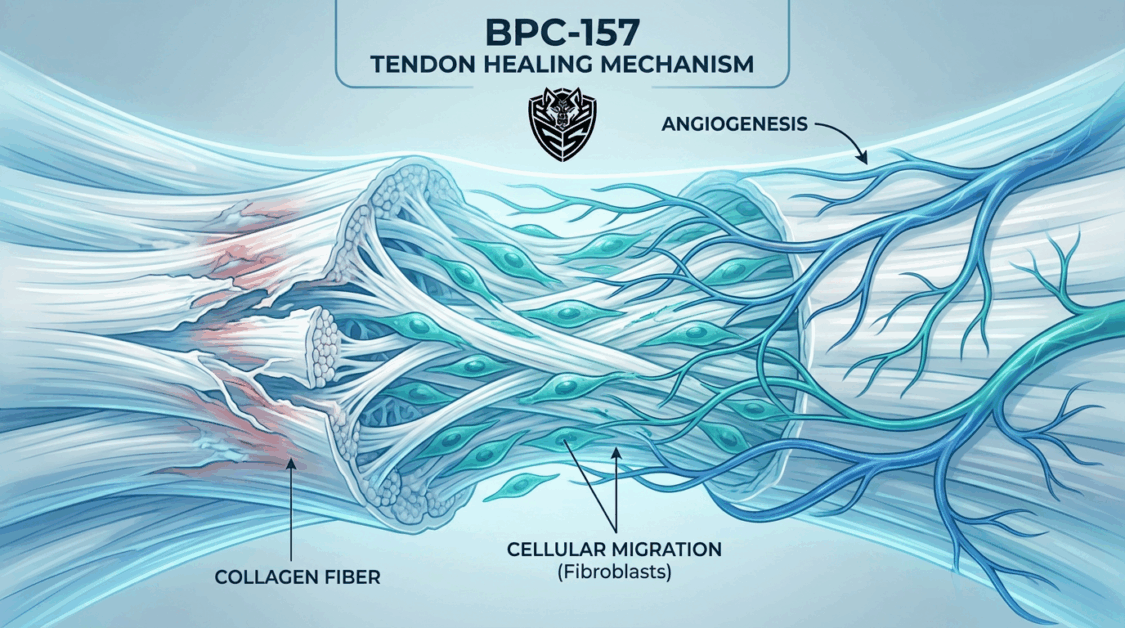
BPC‑157 research shows strong preclinical evidence for accelerating tendon repair by enhancing angiogenesis, fibroblast activity, collagen remodeling, and reducing scar tissue formation. However, human data remain extremely limited, and its use is still investigational.
Musculoskeletal Data Points
- Angiogenesis induction in damaged tissue.
- Upregulation of growth factor receptors.
- Stability in gastric juice for oral research models.
🔬 Mechanisms of Action in Tendon Repair
- Angiogenesis: BPC‑157 upregulates VEGF (vascular endothelial growth factor) and nitric oxide signaling, improving blood supply to poorly vascularized tendon tissue.
- Fibroblast Proliferation: Stimulates tenocyte (tendon fibroblast) migration and collagen synthesis, critical for tendon matrix repair.
- Collagen Remodeling: Promotes transition from Type III collagen (scar tissue) to Type I collagen (strong tendon fibers), improving tensile strength.
- Anti‑Inflammatory Modulation: Balances cytokine activity (↓ TNF‑α, ↓ IL‑6) to reduce excessive inflammation while allowing normal healing.
- Neuromuscular Stabilization: Helps restore function at the tendon‑bone interface and neuromuscular junctions.
📊 Preclinical Research Findings
- Achilles Tendon Models (rats):
- Faster collagen deposition and better fiber alignment.
- 40–70% higher tensile strength compared to untreated controls.
- Accelerated functional recovery (≈40% faster).
- Rotator Cuff Models:
- Improved tendon‑bone healing (enthesis).
- Reduced re‑tear rates in animal studies.
- Patellar Tendon Studies:
- Enhanced collagen maturation (higher Type I/III ratio).
- Better integration of repair tissue with native tendon.
- Ligament Data (MCL):
- Faster healing and improved biomechanical strength, suggesting broader connective tissue benefits.
⚖️ Benefits Observed
- Accelerated Healing Timeline: Shortens recovery phases by weeks.
- Improved Tissue Quality: Stronger, more organized collagen fibers.
- Reduced Scar Formation: Better mechanical properties post‑repair.
- Enhanced Vascularization: More blood vessels in injured tendon zones.
⚠️ Limitations & Risks
- Human Data: Only a handful of pilot studies exist; no large‑scale tendon trials.
- Regulatory Status:
- FDA (2023): Classified BPC‑157 as a Category 2 bulk drug, restricting compounding.
- WADA (2022): Banned in sports under “Unapproved Substances.”
- Safety: Animal studies show no major toxicity, but long‑term human safety is unproven.
- Availability: Widely sold online but outside regulated pharmaceutical channels.
📚 Educational Insight
BPC‑157 is one of the most promising investigational peptides for tendon repair research, with robust animal data showing faster healing, stronger tissue, and improved vascularization. Yet, without controlled human trials, it remains experimental and should be approached cautiously in research contexts.
✅ In summary: BPC‑157 accelerates tendon healing in preclinical studies by boosting angiogenesis, fibroblast activity, and collagen remodeling, but human evidence is minimal and regulatory bodies classify it as investigational.
Would you like me to also prepare a comparison guide (BPC‑157 vs TB‑500 vs PRP) to show how different tendon repair interventions stack up in terms of mechanism, evidence strength, and recovery outcomes?







One thought on “BPC-157 Research Benefits for Tendon Repair”