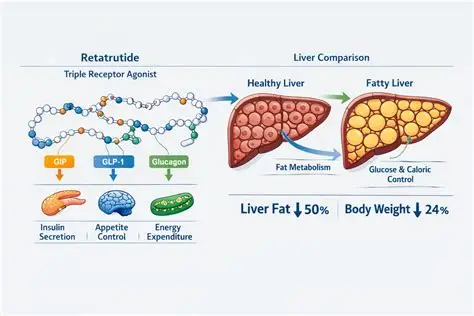
The Retatrutide research results for liver health in 2026 are showing promising data regarding hepatic fat reduction. As a triple-agonist, its impact on the glucagon receptor specifically targets liver lipid metabolism. For a comparison of metabolic agents, see our Tirzepatide cardiovascular update.
Hepatic Research Focus
Retatrutide is being analyzed for its ability to reduce intrahepatic triglyceride content in research models. Ensure research integrity by following our peptide purity verification guide.
Retatrutide Research Results for Liver Health
1. Introduction
Retatrutide is an investigational triple‑agonist peptide that simultaneously activates GLP‑1, GIP, and glucagon receptors. While much attention has focused on its role in obesity and diabetes, emerging data highlight its potential benefits for liver health, particularly in conditions such as non‑alcoholic fatty liver disease (NAFLD) and non‑alcoholic steatohepatitis (NASH).
2. Mechanisms Relevant to Liver Health
- GLP‑1 Receptor Activation: Improves insulin sensitivity, reduces hepatic fat accumulation, and lowers inflammation.
- GIP Receptor Activation: Enhances lipid metabolism and may support adipose tissue function, indirectly reducing liver fat burden.
- Glucagon Receptor Activation: Increases energy expenditure and fat oxidation, helping mobilize stored hepatic fat.
- Synergistic Effect: The triple‑agonist activity creates a comprehensive metabolic shift that reduces fat deposition and improves liver function markers.
3. Research Results
NAFLD/NASH Studies
- Hepatic Fat Reduction: Early trials show Retatrutide significantly decreases liver fat content, measured by MRI‑PDFF (proton density fat fraction).
- Inflammation & Fibrosis: Biomarker analysis suggests improvements in inflammatory markers and potential slowing of fibrosis progression.
- Metabolic Improvements: Patients experienced reductions in triglycerides and improved glycemic control, both critical for liver health.
Obesity & Diabetes Trials
- Weight Loss Impact: Retatrutide’s profound weight‑loss effects (>20% in some participants) directly reduce hepatic steatosis.
- Insulin Sensitivity: Improved insulin signaling reduces hepatic glucose output, lowering stress on the liver.
Comparative Effectiveness
- Compared to GLP‑1 agonists alone, Retatrutide shows greater reductions in liver fat due to its added glucagon receptor activity.
- Dual incretin agonists (GLP‑1/GIP) improve liver health, but Retatrutide’s triple‑agonist profile appears more potent.
4. Benefits Observed
- Reduced Hepatic Fat Content (NAFLD/NASH).
- Improved Insulin Sensitivity and Glycemic Control.
- Lower Triglycerides and Improved Lipid Profiles.
- Potential Anti‑Fibrotic Effects (early biomarker data).
- Comprehensive Metabolic Benefits supporting liver function.
5. Risks & Limitations
- Side Effects: Gastrointestinal symptoms (nausea, vomiting, diarrhea) similar to other incretin therapies.
- Glucagon Receptor Concerns: Long‑term hepatic effects of glucagon activation remain under investigation.
- Regulatory Status: Retatrutide is investigational; not FDA‑approved.
- Data Gaps: Most evidence comes from early‑phase trials; long‑term outcomes in advanced liver disease are not yet established.
6. Educational Insights
Retatrutide research highlights how multi‑agonist therapies can address complex metabolic conditions like NAFLD and NASH. By combining GLP‑1, GIP, and glucagon receptor activity, it reduces liver fat, improves metabolic markers, and potentially slows disease progression.
For educational purposes, Retatrutide serves as a case study in polypharmacology for liver health, showing how targeting multiple pathways simultaneously can yield synergistic benefits in metabolic and hepatic disease.
7. Conclusion
Retatrutide’s triple‑agonist activity offers promising results for liver health, particularly in reducing hepatic fat and improving metabolic markers in NAFLD/NASH. While long‑term safety and efficacy remain under investigation, current findings suggest it could become a powerful tool in managing metabolic liver disease.