The Tirzepatide research results for metabolic health continue to set new benchmarks in 2026. As a dual GLP-1 and GIP receptor agonist, its data points often show a different profile than single agonists. You can see how this compares to newer compounds in our Retatrutide vs Semaglutide analysis.
Dual-Agonist Mechanism
By stimulating two pathways, Tirzepatide provides a comprehensive look at glucose-dependent insulinotropic polypeptide activity. For sourcing, see our guide on where to buy Tirzepatide peptides.
Tirzepatide Research Results for Metabolic Health
🔬 Mechanism of Action
Tirzepatide is a dual incretin agonist that activates both:
- GLP‑1 receptors → reduces appetite, slows gastric emptying, improves glycemic control.
- GIP receptors → enhances insulin secretion, improves lipid metabolism, and supports adipose tissue function.
This dual pathway provides broader metabolic benefits compared to single‑agonist therapies.
📊 Clinical Trial Findings
- SURPASS Trials (Type 2 Diabetes):
- Significant reductions in HbA1c (−2.0 to −2.5%).
- Weight loss up to 20% in many participants.
- Improved insulin sensitivity and beta‑cell function.
- SUMMIT Trial (Obesity & Heart Failure with Preserved Ejection Fraction – HFpEF):
- Reduced visceral adipose tissue (VAT) and improved cardiac remodeling.
- Lowered markers of myocardial stress and improved exercise tolerance.
- Cardiometabolic Outcomes:
- Decreased systolic blood pressure and triglycerides.
- Improved cholesterol balance (↑ HDL, ↓ LDL).
- Reduced systemic inflammation and improved endothelial function.
⚖️ Benefits Observed
- Glycemic Control: Strong HbA1c reduction in type 2 diabetes.
- Weight Reduction: Sustained and clinically meaningful weight loss.
- Cardiovascular Health: Lower blood pressure, improved lipid profiles, reduced heart failure symptoms.
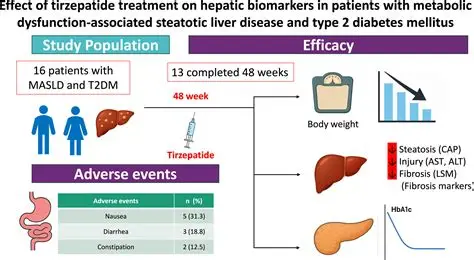
- Liver Health: Evidence of reduced hepatic steatosis (fatty liver).
- Renal Protection: Emerging data suggest stabilization of kidney function.
⚠️ Limitations & Risks
- Side Effects: Gastrointestinal events (nausea, vomiting, diarrhea) are most common.
- Biliary Events: Slightly higher risk at higher doses.
- Pancreatitis: No clear elevation, but monitoring is advised.
- Regulatory Status: Approved for diabetes and obesity; cardiovascular indications under further study.
📚 Educational Insight
Tirzepatide research demonstrates how dual incretin agonism can reshape metabolic health by simultaneously addressing obesity, diabetes, cardiovascular risk, and liver disease. It represents a multi‑target approach that goes beyond glucose control, positioning Tirzepatide as a cornerstone therapy in integrated metabolic care.
✅ In summary: Tirzepatide delivers robust improvements in glycemic control, weight loss, cardiovascular outcomes, and liver health, making it one of the most promising therapies in modern metabolic research.







One thought on “Tirzepatide Research Results for Metabolic Health”